Chemical equations are one of the most fundamental aspects of chemistry, and it’s also one of the most complicated. It’s a science that spans all fields, including physics, math, engineering, biology, and even psychology. The best way to understand chemistry is to start at the beginning and learn how to solve problems.
Chemistry and Chemical Equations Homework Help
This comprehensive guide will teach you everything you need to know about chemistry on all levels. From understanding what the different units mean in chemistry equations to calculating order of reactions and balancing chemical equations, read on for a detailed guide on understanding chemistry.
Chemistry is the science of matter, and it’s one of the most fundamental sciences. It’s a science that spans all fields, including physics, math, engineering, biology, and even psychology. The best way to understand chemistry is to start at the beginning and learn how to solve problems.
This comprehensive guide will teach you everything you need to know about chemistry on all levels. From understanding what the different units mean in chemistry equations to calculating order of reactions and balancing chemical equations, read on for a detailed guide on understanding chemistry.
The study of chemistry is fascinating because it involves so many different concepts like matter, atoms, molecules, reactions, equilibrium reactions, stoichiometry and more! There are also several practical applications which include food science as well as drug discovery. So if you are interested in learning more about this fascinating field then read through this guide today!
Chemistry is a science that spans all fields. If you want to learn more about chemistry and its complexities, one of the first things you need to understand is how to use the different units of measurement in chemistry.
There are three main types of units used in chemistry:
The most common unit used in chemistry is the gram (g). This unit is used to measure mass, volume, and length. Other units include: tons per cubic meter (tonne/m), kilograms per liter (kg/L), kilograms per cubic meter (kg/m), meters per liter, centimeters per liter, milliliters per kilogram, nanoliters per milliliter, and picometers per micrometer. The abbreviation for these units can be found as follows: g, tonne/m, kg/L, kg/m, m/L, mL/kg, nl/mL, pm/μm.
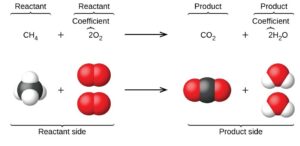
A stoichiometry problem is one in which the goal is to determine the number of moles of a substance that react with other substances. Balancing chemical equations is the process of determining how many moles of a compound are produced during a reaction and what the starting material quantities are.
To balance a chemical equation, start by adding up the amounts of each component in your equation. Subtract out any non-reactants from your equation as well, such as water or air that isn’t involved in the reaction. The answer you get will be in moles. For example, if you have 50 grams of reactant A, 100 grams of reactant B, and 200 grams of product C, then your balanced chemical equation would look like this:
A+B=50
C=100-200
A reaction is the process of change that takes place when two or more substances react. There are several different types of reactions, but the most common types are addition and substitution reactions. Reactions can also be classified as exothermic or endothermic based on whether heat is generated or released during the reaction.
There are three different ways you can calculate order of reactions:
1) The empirical method
2) The colligative property method
3) The Le Chatelier’s principle method.
Chemical equations are the original and most popular way to solve problems in chemistry. Chemical reactions are usually written in chemical equations, so it is important to understand how to write them.
A chemical equation is made up of a chemical substance (reactive) that reacts with something else. The thing reacting is called the reactant or catalyst, and the thing that is not reacting is called the product or goal element.
This equation will have a number of variables, which will be denoted by letters representing certain units. For example, in order for hydrogen gas to react with chlorine gas, each of these gases would have a variable denoted by “H” and “Cl”. These are shorthand for hydrogen atoms and chlorine atoms.
Recent Comments